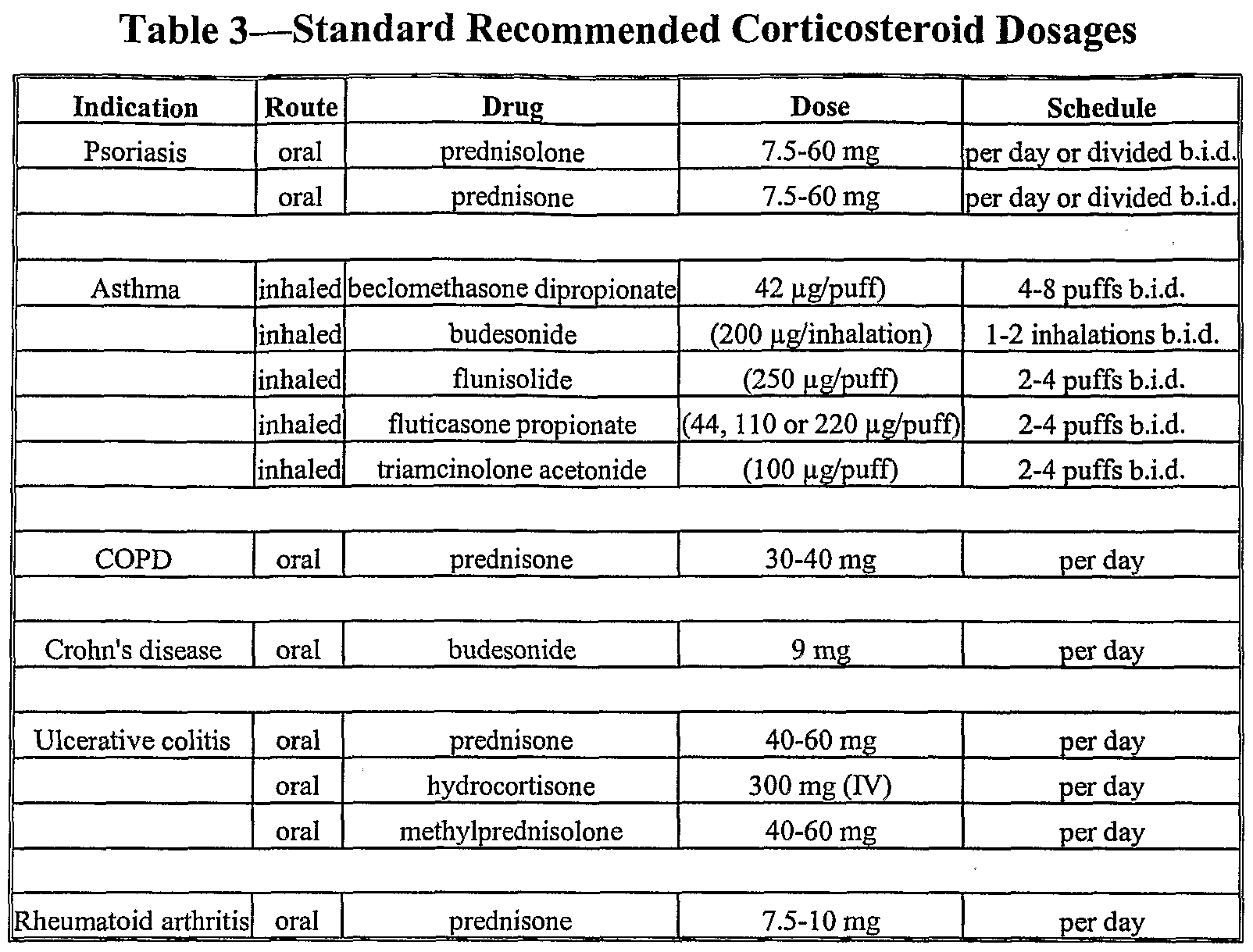
adults with an initial body mass index (BMI) of ≥30 kg/m 2 (obesity) or ≥27 kg/m 2 (overweight) in the presence of at least one weight-related comorbid condition (e.g., hypertension, type 2 diabetes mellitus, or dyslipidemia).Wegovy ® (semaglutide) injection 2.4 mg is indicated as an adjunct to a reduced calorie diet and increased physical activity for chronic weight management in: Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with Wegovy ® a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Counsel patients regarding the potential risk for MTC with the use of Wegovy ® and inform them of symptoms of thyroid tumors (e.g. Wegovy ® is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). 
It is unknown whether Wegovy ® causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined In rodents, semaglutide causes dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
